With the hope of overcoming the generation of hazardous materials to human health and environment, serious and great endeavor have been made in catalyst fabrication using green chemistry technology. In this paper, the manganese (III) acetylacetonate nanoparticles with diameters of about 146 nm were prepared by a simple and environmentally benign route based on hydrolysis of KMnO4 followed by reaction with acetylacetone in rapid stirring rate or ultrasonication. The as-prepared samples were characterized by X-ray diffraction, energy dispersive X-ray fluorescence (EDIX), Fourier transfer infrared spectroscopy and scanning electron microscope. Various parameters were investigated, and the pure and stable crystals of manganese (III) acetylacetonate could be obtained in 98% conversion at a molar ratio 7∶1 of acetylacetone to KMnO4 and 75°C after 60 min. We further proposed a mathematical model, and the predicted results from model were in good agreement with experimental results.
Metal acetylacetonate complexes are widely used as catalyst and co-catalysts in many chemical reactions such as oligomerization, polymerization, isomerization, hydrogenation, and coupling. Also, they are used in rubber technology for vulcanization [ 1, 2].
The unique properties of these coordination complexes have made them multi-field researches such as stereoisomerism, where the tris-chelate complexes occurs due to the fundamental difference in two possible ligand bonding configurations. Likewise, coordination compounds arise when a metal cation is bonded to one or more anions through coordinate covalent bonds [ 3].
According to that, metal acetylacetonates are stated to promise precursor of heterogeneous catalysts [ 4, 5, 6]. Volatility of some acetylacetonates (Cr(acac)3, Al(acac)3, V(acac)3, Pt(acac)2 or Cu(acac)2) makes possible their vapour phase to deposite on support surface. Thermally unstable acetylacetonates (Co(acac)2, Fe(acac)3 or Mn(acac)3) can be deposited by adsorption from organic solvents. High dispersion degree of supported metal acetylacetonetes is achievable due to their H- bonding with surface groups of support or ligand substitution [ 7, 8, 9, 10, 11]. Also, manganese (III) acetylacetonate (Mn(acac)3) has been employed as efficient constituent in electrolyte of single-metal redox flow battery (RFB) for applications including storage of the large amounts of energy produced by intermittent sources such as wind turbines and solar cells. However, Mn(acac)3 was chosen as source material for preparation of LiMn2O4 thin films, which are used as a cathode material in microbatteries [ 7]. However, these compounds can be used in polymeric coatings as stabilizer of cross-linking reaction [ 12, 13, 14], catalyst of drying process [ 9, 10] and toughening agent [ 11].
Green chemistry has attracted more attention in the past century to use non-toxic or less toxic raw materials and solvents to produce environmentally benign chemicals, because its definition is invention, design, development and application of chemical products and processes to reduce or eliminate the use and generation of substances hazardous to human health and environment [ 4, 5, 6]. Metal acetylacetonate complexes can be produced by green chemistry using acetylacetonate anion and metal ions, usually transition metal compounds.
The present study investigates the production of Mn(acac)3 with a green chemistry technique; it does not use any hazardous solvent other than water, in which the product is insoluble and thus can be easily separated. Also, this process uses one source of manganese that has a high oxidizing power to maximize reaction yield, and could be an alternative for non-green traditional technique. Another benefit of this green technique is the high purity of produced Mn(acac)3 due to the absence of any contaminants and easily recycling of unreacted reactants [ 15, 16]. Traditionally, Mn(acac)3 is prepared by non-green technique in the presence of sodium acetate as a neutralizing agent in reaction mixture; the reaction begins with MnCl2·4H2O and acetylacetone to form a complex of Mn(II) which in turn reacts with Mn(IV) from KMnO4 to provide Mn(acac)3. The resulting product will require extra washing and separation techniques before final application. Moreover, sodium acetate, unreacted manganese sources and acetylacetone in the final filtrate are difficult to separate, recycle and reuse [ 17].
The objective of this work is to develop a reliable production technology to produce pure crystals of Mn(acac)3 in nanostructure. Quality control test on the resulted products are introduced to guarantee its promising industrial application as an efficient nano-scale catalyst. Also, this work provides an improved, economical and environmentally benign green process for the development of Mn(acac)3
Technical grade acetylacetone was purchased from Fluka, potassium permanganate (KMnO4) was obtained from Sigma-Aldrich. Chemicals were used as received unless stated otherwise.
Mn(acac)3 was synthesized by dissolving 5 g of KMnO4 in 50 mL of distilled water with continuous stirring in batch system, while the heating of reaction was carried out by a water bath. After dissolution was completed; distilled acetylacetone was added to the solution with continuous stirring. A white crystalline precipitates were observed during the reaction. Therefore, it was important to keep the whole mixture to cool for 10 minutes in order to complete the crystals deposition. The formed dark shiny crystals of Mn(acac)3 was filtered off and dried in vacuum over fused CaCl2 for 15 minutes.
A parametric study was conducted to investigate the influence of molar ratio of acetylacetone to KMnO4, reaction temperature, and reaction time. Production of Mn(acac)3 was accomplished without any neutralizing agent to prevent contamination so the product purity and stability upon storage increased [ 17, 18, 19, 20].
A variety of instrumentation analyses were performed to detect and evaluate the quality of final product (Mn(acac)3 crystals), allowing the formulation of an integrated full product assay by Fourier transfer infrared spectroscopy (FTIR), scanning electron microscope (SEM), energy dispersive X-ray fluorescence (EDIX) and X-ray diffraction (XRD).
After the reaction was completed, the produced solution was filtered to separate the dark shiny crystals of Mn(acac)3 from unreacted solutions. Then, the unreacted manganese in reaction solution was analyzed by HPLC Agilent 1200 series, made in Germany, using methanol as mobile phase and Eclipse XDB-C18 (5 μm, 4.6 × 150 mm) as the stationary phase. Finally, the reaction conversion can be calculated according to this formula:
Where x is the reaction conversion, Mnin is the moles of inlet manganese according to KMnO4 amount, and Mnunreacted is the unreacted moles of manganese which was obtained from HPLC analysis of produced unreacted solution.
Mn(acac)3 crystals samples were grinded and coated with gold sputtering to provide electrical conductivity. The micrographs were taken on a JEOL 5410 scanning electron microscope at 20 kV. EDIX was used to measure the elemental analysis of Mn(acac)3 crystal samples, using the energy-dispersive X-ray fluorescence (EDIX) device contains DET, area-10 mm2, window (ATW2) model 6587, made in Oxford.
Samples of Mn(acac)3 crystals were also characterized by FTIR analysis at 16 scans per minute with resolution of 4 cm-1 and absorbance 400-4000 cm-1. The IR data were collected using JASCO FTIR-6100 model, made in Japan.
XRD analysis was used to identify the mineralogical composition of Mn(acac)3 crystal samples. The XRD-spectrum (Brukurd 8 advance, CuK, target with secondary mono chromator λυ = 40, mA= 40, Germany) was used for Mn(acac)3 crystals identification and estimation of the anastase-to-rutile ratio. XRD patterns were obtained at room temperature by using X-ray generator (Shimata XD-D1, Japan).
Mathematical model was developed using MATLAB Simulink (Mathworks, Release 2008a) and the batch processes were studied with respect to different parameters (molar ratio, temperature, and reaction time). The model was verified by comparing experimental results with predicted results from the model.
A series of experiments were carried out using different molar ratios (1 ∶ 1 to 10 ∶ 1) of acetylacetone to KMnO4. The other conditions were fixed at 45°C and 60 min for reaction time. Samples were taken from the reactor flask after 10 min during the reaction time. Fig. 1 indicates unreacted Mn% in produced filtrate solution, where the lowest weight percent of Mn in the filtrate was 1.74% at molar ratio 10 ∶ 1 of acetylacetone to KMnO4 which increases to be 1.79% and 1.84% at molar ratios 7 ∶ 1 and 5 ∶ 1 respectively. It was obvious from these experiments that excess molar ratio between the reacting species greatly affects the reaction conversion and product yield as shown in Table 1. The molar ratio 7 ∶ 1 other than 10 ∶ 1 was chosen to be optimum because the corresponding reaction conversions are approximately close to each other.
| Tab.1 Effect of various molar ratio on reaction conversion of Mn(acac)3 production after 60 min |
To further optimize the reaction conditions, different reaction times (5 min, 15 min, 25 min, 35 min, 45 min and 60 min) were tested. In these experiments, the other reaction conditions were fixed at molar ratio 7 ∶ 1 of acetylacetone to KMnO4 and at 45ºC. Figure 2 illustrates yield of Mn(acac)3 at various reaction time, where the yield reached 7.5 gm after only 5 min and increased to 10.62 gm after 60 min. Increasing the yield during the reaction time is common in reactions of strong inorganic oxidizing reagents with organic compounds [ 16] and this was also verified with the following mathematical model. The optimum reaction time was 60 min, which was long enough for the optimization process by minimizing the unreacted compounds and increasing process economic.
In order to use less energy and reduce the chemical reaction costs, experiments were carried out at 25°C, 45°C, 55°C, 65°C and 75°C, respectively, with a 7 ∶ 1 molar ratio of acetylacetone to KMnO4 and reaction time being 60 min. Figure 3 reveals that unreacted Mn% in filtrate liquid phase is always decreased to reach a minimum of 0.49% at 75ºC compared with 1.79% at 45ºC.
These results indicate that by increasing reaction temperature the reaction conversion could be increased to 94% at reaction temperature 45ºC and about 98% at 75ºC. However, increasing temperature (up to 75ºC) could cause excessive control on reaction vessel design due to accessibility to foam formation at higher temperature, and increase operating cost of required energy consumed.
Mn(acac)3 crystals precipitated from solution was subjected to full chemical analysis to confirm its structure and purity. Scanning electron micrographs were used for Mn(acac)3 crystal surface views with the magnification 120000 and 100000 respectively in the micrographs (a) & (b) as shown in Fig. 4. The SEM images indicate that Mn(acac)3 was uniform fine particles with particle size of 146 nm and wall thickness 60 nm without using sophisticated experimental technique in mixing and ultrasonication, which could lead to the formation of crystalline aggregates of this product. This means that slight modifications using ultrasonic homogenizer can form small nano-particles of this product which can be applied in catalysis of chemical processes.
 | Fig.4 SEM micrographs of(a) crystal size of manganese (III) acetylacetonate, and (b) wall thickness of manganese (III) acetylacetonate crystals |
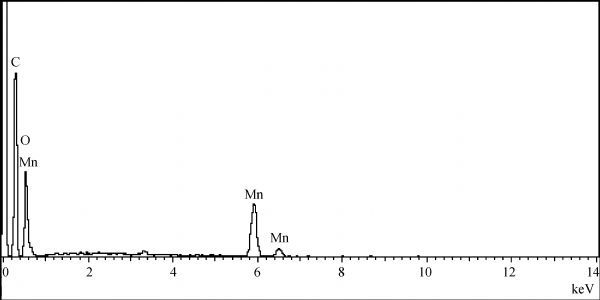
Also, the metallic content of Mn% in solid crystals of product was 14.7%, as confirmed by EDXRF, whereas the organic carbon content was 50.21% as analyzed by the elemental analysis Fig. 5 showed the EDIX chart for produced crystals, agreeing with the published literature [ 19]. The result from HPLC was 14% Mn, which is also in agreement with analysis in previous literatures [ 20, 21, 22].
The produced Mn(acac)3 crystals were characterized by FTIR. Its FTIR-spectrum (Fig. 6) was super-imposable on that of the authentic sample particularly in the figure print region (1500-650 cm-1). The main distinguishing features were: (i) the presence of strong absorption bands (peaks nos. 14, 15, and 16) at 1588, 1514.8, and 1384.6 cm-1 respectively (C–O, carboxylate), (ii) peak no. 23 at 753.5 cm-1, and (iii) peak no. 26 at 565 cm-1 (Mn–O, stretching). Fig. 5 proves excellent agreement with published literature [ 21].
Figure 7 illustrates the XRD-spectrum that reflects the amorphous state of the final product nano-molecules. The 2nd and 3rd peaks ( d = 5.877 and d = 5.48040) appear correspond to the associated organic impurities, while the 1st major sharp peak ( d = 8.05609) belongs to the main product. The developing XRD-monograph is really considered as figure-prints for product identification. However, the XRD results were in agreement with the published literature [ 23].
The overall reaction equation was
| (1) |
In order to obtain kinetic equation of the reaction, the experimental results were correlated to kinetic model for the reaction which was carried out in a glass batch system of 200 mL capacity. Therefore, the feed of reaction was charged to reactor and then heated to the desired reaction temperature which was kept constant within±0.5 K.
The kinetic reaction was conducted by withdrawing samples at regular time intervals that were analyzed by HPLC to determine unreacted KMnO4 and thus reaction conversion.
Assumptions used to develop the model included:
• The reaction rate was defined as the change in the rate of moles for component i with time gmol·sec-1. The rate of reaction was expressed in terms of the moles of the different components.
• The reaction was considered pseudo first order reaction, where the acetylacetonate was used in excess amount (acetylacetone /KMnO4).
• KMnO4 was used as oxidizing agent, where the permanganate ion
• The presence of excess acetylacetone can reduce
• Increasing amount of produced water can dissolve the byproducts and reverse the reaction. In aqueous solution, Mn (III) can readily hydrolyze according to Eq. (2) but it is most stable in acid solutions. Manganese (III) is also slowly reduced by water as in Eq. (3).
| (2) |
| (3) |
So, the molar ratio of acetylacetonate to KMnO4 must be high enough to keep acetylacetonate concentration constant throughout the process, where using excess amount of acetylacetonate can break thermodynamic equilibrium of the reversible reaction according to LeChâtelier principle [ 24].
The reaction rate constant ( k) as function of temperature can be expressed by the Arrhenius equation.
| (4) |
Equilibrium constant ( K) is also function of temperature and can be expressed by Van’t Hoff equation [ 25].
| (5) |
| (6) |
| (7) |
| (8) |
The rate of reaction is calculated according to overall reaction equation (9):
| (9) |
The reacted amount of KMnO4 and the produced amount of Mn(III) acetylacetonate can be calculated from Eqs. (10), and (11), respectively.
| (10) |
| (11) |
The reaction conversion can be calculated from Eq. (12).
| (12) |
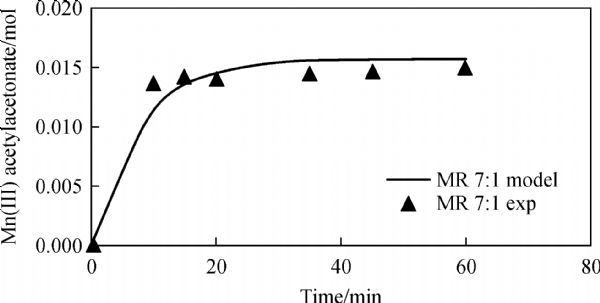
The effect of molar ratio of acetylacetone to KMnO4 on the reaction conversion was investigated at 1 ∶ 1, 2 ∶ 1, 4 ∶ 1, 5 ∶ 1, 7 ∶ 1, and 10 ∶ 1, respectively. The results show that the unreacted Mn decreased as the molar ratio increased. However, molar ratio 7 ∶ 1 was chosen for the highest conversion (94%) after 60 min at 45°C which were close to the reaction conversion at molar ratio 10 ∶ 1. The comparison between predicted unreacted Mn from model and experimental results using molar ratio 7 ∶ 1 is shown in Fig. 8, indicating a good match with experimental results. The heat of reaction was calculated to be -4336 J/mol, the change in Gibbs free energy was -7326.6 J/mol and entropy change was 9.4 J/mol·K. Table 2 illustrates parameters calculated by model.
 | Fig.8 Comparison between predicted unreactant with model and experiment results (molar ratio of acetylacetone ∶ KMnO4 = 7 ∶ 1) |
| Tab.2 The model parameter at different molar ratios |
Figure 9 illustrates comparison between predicted produced Mn(acac)3 moles from model and experimental results using molar ratio 7 ∶ 1 of acetylacetone to KMnO4, indicating the experimental results fitted with the predicted model.
The effect of temperature on the rate of reaction was studied by conducting the reaction at 25ºC, 45ºC, 55 ºC, 65 ºC and 75ºC, respectively. The molar ratio of acetylacetone to KMnO4 was fixed at 7 ∶ 1 with reaction time 60 min. The results show that the reaction conversion increases as the temperature increases, thus the productivity of Mn(acac)3 increases. Fig. 10 illustrates comparison between predicted results and experimental results according to reaction conversion at temperature 75°C, showing a fair match between predicted model results and experimental data. Model parameters at various temperatures were calculated and illustrated in Table 3. The change in heat of reaction change, Gibbs free energy and change in entropy were calculated at different temperatures.
 | Fig.10 Comparison between predicted model of produced Mn(acac)3 and experimental results at 75°C (molar ratio of acetylacetone : KMnO4 = 7 ∶ 1) |
| Tab.3 Model parameters at various temperatures |
When the kinetic model was fitted to all experimental results, the results were in good agreement.
The cumulative productivity of reaction was studied at reaction temperature 75°C, molar ratio 7 ∶ 1. Figure 11(a) demonstrates the cumulative productivity over 120 min of operation. Figure 11(b) depicts the slope of the curve, which shows that the maximum production rate of 0.000732 mol/ min was reached after 20 min. Therefore, the optimum time to shift a new cycle can occur after 20 min. The scaling up of the reaction can be suggested to use stainless steel batch reactor with three openings one for condenser, one for feeding at the top of the reactor and the last one for collecting product at the bottom of the reactor; this can provide in a concurrent increase in the reactor area from 200 mL to 10 L. The reactor must have temperature controller and pressure gauge for safety. However, increasing in reactor volume means high cumulative productivity thus increasing in the product yield. The system can be connected to filtration system to filter the product from unreacted reactants and byproducts.
 | Fig.11 (a) Change of cumulative productivity with time at 75°C and molar ratio 7 ∶ 1; (b) The production rate versus time at 75°C and molar ratio 7 ∶ 1 |
We have demonstrated that Mn(acac)3 nanoparticles could be synthesized in aqueous medium through chemical precipitation technique. Under the following conditions: molar ratio 7 ∶ 1 of acetylacetonate to KMnO4, reaction temperature 75°C, and reaction time 60 min, the highest reaction conversion of about 98% was obtained.
The final product was analyzed by FTIR, SEM, EDIX and XRD, and all analyses proves excellent agreement with published literature [ 19]. The SEM images showed that Mn(acac)3 was obtained as uniform fine nanoparticles with particle size of 146 nm and wall thickness 60 nm without using sophisticated experimental technique. The mathematical model of the reaction was verified and adjusted based on experimental results.
Nomenclature
| K | ate reaction constant (gmol/min), |
|---|---|
| A | requency factor |
| Ea | ctivation energy (J/mol) |
| R | as constant (J/mol·K) |
| T | eaction temperature (K) |
| Δ H | hange of heat of reaction (J/mol) |
| ΔG | hange of Gibbs free energy of reaction (J/mol) |
| Δ S | ntropy change of reaction (J/mol·K) |
| K | quilibrium reaction constant |
| K0 | quilibrium reaction constant at room temperature 25°C |
| 1 | MnO4 moles (gmol) |
| | n(III) acetylacetonate moles (gmol) |
| X | eaction conversion |
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
| 10 |
|
| 11 |
|
| 12 |
|
| 13 |
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
|
| 18 |
|
| 19 |
|
| 20 |
|
| 21 |
|
| 22 |
|
| 23 |
|
| 24 |
|
| 25 |
|