The composite separator comprising of polysulfone and zirconia was prepared by phase inversion precipitation technique. The influence of manufacturing parameters on its properties was investigated, and the results show that the manufacturing parameters affect the ionic resistance and maximum pore size significantly. A modified composite separator with a support layer was prepared to enhance the tensile strength of separator. By adding support layer, the tensile strength of the separator increases from 1.85 MPa to 13.66 MPa. In order to evaluate the practical applicability of the composite separator, a small-scale industrial electrolytic experiment was conducted to investigate the changes of cell voltage, gas purity and separator stability. The results show that the modified composite separator has a smaller cell voltage and a higher H2 purity than the asbestos separator, and are promising material for industrial hydrogen production.
With increasing concern for the depletion of fossil fuels and environment problems, hydrogen energy has been widely considered as a promising and significant substitution of fossil fuels. After adequate investigation, alkaline water electrolysis has become a mature technology with several advantages, such as its easy operating and high hydrogen purity. However, this technology still has some aspects to be improved, for instance, the cost of hydrogen production by this means is higher than that from fossil fuels [ 1].
Operational voltage of the electrolyzer, which is related to the separator property, directly affects the cost. Asbestos has been widely used as the main material of alkaline water electrolysis separator in the last century. However, it is toxic and carcinogenic [ 2, 3]. For this reason, much effort has been made to find a new diaphragm material to replace it.
An ideal separator should have the following properties: (i) high chemical and mechanical stability in strong alkaline medium, (ii) high porosity and small pore size, (iii) good wettability allowing ionic transport in the solution between electrodes, and (iv) low gas permeability. Because of their excellent thermal and chemical stability, uniform pore size and long service life, inorganic separators are promising materials for high temperature gas-separation. It has been reported [ 4] that NiO diaphragms with a thickness of only 0.2 mm exhibit excellent chemical and mechanical stability, as well as good gas-separation property and corrosion resistance in high temperature alkaline medium. Devisek and Mergel [ 5] found that adding 5 wt-% TiO2 into NiO diaphragm effectively prolongs its service life. Takashi and co-workers [ 6] have developed a novel method to fabricate the compact NiO diaphragms with the controllable thickness and porous structure. However, H2 produced can deoxidate NiO under such conditions, which limits its application in alkaline water electrolysis.
With the development of high polymer material, investigators have been trying to use organic materials to prepare separators. An organic diaphragm has been prepared [ 7] from polyphenylene sulfide (PPS) fiber after hydrophilic treatment, and exhibits high mechanical strength and corrosion resistance in 30 wt-% KOH solution under 200 atm pressure. However, its average pore size of 32.8 μm is difficult to meet the operating requirement, and the purity of H2 is too low. Zhuang and co-workers [ 8] have prepared a homogenous blend membrane from poly ether sulphone (PES) and poly vinylpyrrolidone (PVP), which exhibits good chemical and mechanical stability. Both inorganic and organic separators have their own advantages and weaknesses. A novel composite separator with a combination of their advantages would meet the rigorous requirements for alkaline water electrolysis. Vermeiren and co-workers [ 9, 10, 11] have prepared Zirfon® separator (ZF) composed of ZrO2 powders and polysulfone (PSF). ZrO2 powder is hydrophilic and has a high specific area, which assure an optimum wettability. PSF renders the separator the enough mechanical strength. The resistivity of the separator with a thickness of 628 μm was only 3.25 Ω·cm in 30% KOH solution. In addition, the corrosion resistance and ionic conductivity of the separator are excellent. The authors have also investigated the influence of manufacturing parameters on the properties of Zirfon®, however, the investigation has not been consummated. Besides, we found that the mechanical strength of ZF separator is lower than asbestos. Without this defect, the ZF separator could be an optimum substitution for asbestos.
In this work, to overcome the defect of the ZF separator, we have investigated the effects of manufacturing parameters on its mechanical strength and other properties. Furthermore, we have prepared a modified ZF separator with a support layer, and have also studied its properties.
The manufacturing procedure for porous separators is based on the phase inversion precipitation technique. At first, polysulfone, zirconia and polymeric additive (PVP) with different concentrations were dissolved in N-methy1-2-pyrrolidone (NMP) to prepare the polymeric solution, which was then cast onto a glass sheet and spread by a scraper to form a film with constant thickness. The resulting film was exposed to air for constant seconds, and then the glass sheet was immersed in a non-solvent bath which contained deionized water at constant temperature. After a few minutes, the obtained separator was rinsed several times with deionized water and then was stored in deionized water for further characterization.
The separators with the support layer were prepared by the same procedure as above except that the support layer was immersed into the casting solution sufficiently before being spread. The material of support layer was PPS with different density (Separator A: 99 g·m-2, B: 122 g·m-2, C: 152 g·m-2 and D: 198 g·m-2).
In this work, all the separators prepared comprised of 60 wt-% ZrO2 and 40 wt-% PSF. Other preparation conditions are as follows: PSF/PVP/NMP is 15%/15%/70%, the pre-evaporation time is 15 seconds, and the extraction temperature is 15 °C unless otherwise stated.
Different characterization techniques were used to test the properties of the separators. The maximum pore size was obtained by bubble-point (BP) method. In this method, the relationship between the maximum pore size and BP pressure is given by Laplace Eq. (1):
where d is the diameter of the largest pore; g is the surface tension at the liquid air interface; q is the contact angle of alcohol with the separator material; and D P is the pressure difference applied across the separator. BP pressure is considered as one of the most important properties of a separator [ 10]. In alkaline water electrolysis, this BP pressure should be high enough to avoid mixing of H2 and O2 generated in the electrolyzer. Therefore, a small pore size is desired based on this equation. In our test, the BP pressure was determined in a test cell where the top of the separator was covered with alcohol and the bottom of the separator was pressed by the air with increasing pressure. The BP pressure was corresponding to the first air bubble penetrating through the separator.
The ionic resistance was measured in an electrolysis set up like Fig. 1 at 25 °C, where the electrolyte solution was 30 wt-% KOH. Before testing, the separator was immersed into the absolute ethyl alcohol for 2 h, and then into 30 wt-% KOH solution for 24 h. The ionic resistance was calculated by the following equations:
where I is the current density (A·m-2); i is the value of ampere meter (A); s is the effective area of the separator (m2); R m and R 0 are the ionic resistance corresponding to the electrolyser with and without separator respectively (W·m2); and U is the value of voltmeter.
The ionic resistivity is related to the ionic resistance and separator thickness. The resistivity of the separator was calculated according to the Eq. (4):
where r is the ionic resistivity (W·m), and d is the separator thickness in wet state (m).
The tensile strength in this paper was calculated according to the national standard GB1039-79 and GB1040-79 by the following equation:
where σ is the tensile strength (MPa), P is the maximum loading when the separator break (N), d is the separator thickness (mm), and b is the width of the separator.
The porosity of the separator was tested by weighting method, and calculated according to the following equation:
where ϵ is the porosity of the separator (%), mw and md are the mass of the separator in wet and dry (g) respectively, ρ is the density of deionized water (g/mL), A is the area of the separator (cm2), and d is the separator thickness in wet (cm).
Corrosion resistance was obtained according to the ministry of construction standard JC211-77 “membrane asbestos cloth standard”. Impact resistance was obtained by the same method, except that the mass loss of the separator included both the corroded and destroyed, whereas in corrosion resistance the destroyed mass loss was neglected.
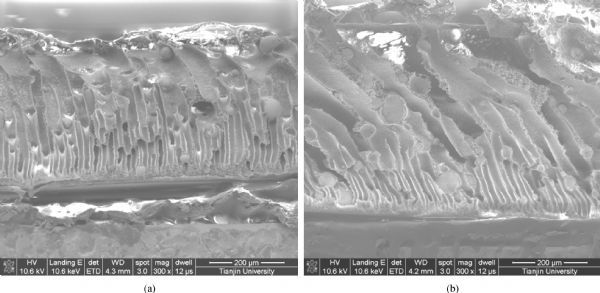
Systematic thermodynamics and kinetics have great effect on the performance and structure of separators fabricated by the phase inversion precipitation technique [ 12]. The formation of pores is determined by the speed of nucleation and the speed of polymer-poor phase growth [ 13, 14]. The separator thickness can directly affect the growth of the polymer-poor phase. Therefore, the thickness of wet film has a great impact on the structure of the pores inside the film formed by phase inversion precipitation technique [ 15]. Four separators with different thickness (about 150 μm, 300 μm, 450 μm and 600 μm) were tested and compared.
Table 1 gives an overview of several characterization tests. As can be seen in Table 1, the ionic resistance increases significantly when the thickness rises from 150 μm to 600 μm. However, the increasing is not linear, and instead the value is less than that expected when the thickness is greater than 150 μm, because both porosity and thickness affect the ionic resistance. With increasing thickness, the pore size increases, and the tortuosity of the separators decreases [ 11], so the ionic resistance was reduced.
When the wet film is not thick enough, polymers on the film surface shrink severely due to the evaporation of solvent during pre-evaporation process, and dense layers could be formed easily. However, the layers tend to be loose when there is a huge increment in thickness. Table 1 shows that there is a slight increment in pore size as the thickness increases. Gas permeation properties of separator are directly affected by the separator structure [ 16]. The influence of thickness on gas separation performance is greater, as the thicker barrier helps to prevent gases from mixing. Therefore, the gas separation performance of the separator may be improved as the thickness increases.
| Tab.1 The effect of thickness on the properties of ZF separator |
When the separator becomes thicker, the connections among pores decrease. Fig. 2 shows the cross-section images of composite separator with different thickness. Although Table 1 shows that the tensile strength declines slightly as the thickness increases, thicker separators are not damaged during separator installment; hence thicker separators are favored by operators. The alkali-corrosion loss remains unaffected by thickness.
The pore size is directly affected by the polymer concentration in the solution. In this section, four separators with different PSF concentrations (10 wt-%, 12 wt-%, 15 wt-% and 20 wt-%) were compared. The influence of PSF concentration on the properties of ZF separator is listed in Table 2.
| Tab.2 The effect of PSF concentration on the properties of ZF separator |
As can be seen in Table 2, the maximum pore size and porosity decrease as PSF concentration increases, probably because the space among polymer is decreased (Fig. 3), and thus both the cortical surface and the inside of the separator become denser. Meanwhile, the viscosity of the solution is increased, which causes a delayed mutual diffusion between the solvent in the casting solution and the non-solvent, and concomitantly inhibits the pore forming. The ionic resistance is increased significantly due to the decreasing porosity and increasing thickness. However, the tensile strength is stronger because the higher concentration of PSF is beneficial for forming connections among PSF molecules, and thus the binding force is strengthened. In addition, the porosity and thickness can both affect the gas separation performance. The declined porosity and increased thickness would make it harder for gas to pass the separator so higher PSF concentration may benefit gas separation. The alkali-corrosion loss of these four separators remains stable (from 0.1% to 0.2%) due to the excellent corrosion resistance of PSF and zirconia.
On one hand, the addition of PVP increases the viscosity of casting solution and inhibits the mutual diffusion between the solvent and the non-solvent. On the other hand, PVP also influences the thermodynamics stability, and is beneficial to demixing [ 17]. Thus, the influence of PVP on the demixing property of ZF separators is complicated. In this section, five separators with different PVP concentration (0%, 5%, 10%, 15% and 20%) were tested. Table 3 gives an overview of several characterization tests.
| Tab.3 The effect of PVP concentration on the properties of ZF separator |
Another important role of PVP is porogenic. Because PVP is hydrosoluble, when the separator is put into deionized water, PVP quickly dissolves to generate pores. As shown in Table 3, the maximum pore size and porosity increases. Also from Fig. 4, it can be seen that the separator tends to be loose with the PVP concentration increasing so the tensile strength decreases. Furthermore, the loosened structure of the separator is easily damaged in the alkali-corrosion test, and thus the mass loss of the separator is increased.
The porosity directly affects the ionic resistivity because there is no significant difference among those separators in thickness. As can be seen in Fig. 5, the ionic resistivity decreases owing to the increased porosity. And the gas separation performance would be declined with the increase of maximum pore size and porosity.
 | Fig.4 SEM cross-section images of membrane with different PVP content: (a) 0%, (b) 5%, (c) 10% and (d) 20% |
Because ZrO2 has excellent hydrophilia, the hydrophilia of the separator could be improved by adding ZrO2 [ 18]. The addition of ZrO2 also affects the morphology and properties of the separator. In this section, nine separators with different ZrO2 concentration (0%, 10%, 20%, 30%, 40%, 50%, 60%, 70% and 80%) were prepared and tested, and the results are summarized in Table 4.
| Tab.4 The effect of ZrO2 concentration on the properties of ZF separator |
The addition of ZrO2has two opposite influences on the pore diameter and structure. On one hand, because ZrO2is a hydrophilic inorganic particle, it can promote the exchange of solvent and water, and thus increase the porosity and pore size. On the other hand, as shown in Table 4, the addition of ZrO2 increases the viscosity of cast solution, which would inhibit the formation of large pores and lead to form a dense surface layer [ 19]. As shown in Table 4 and Fig. 6, the variation of pore size and porosity with different ZrO2 concentration seems to be complicated. Anyway, the dense structure has good effects on corrosion resistance, and makes the separators more fastness.
The ionic resistivity is decreased due to the improvement of hydrophilia by the addition of ZrO2particles. Vermeiren and co-workers have also reached a similar conclusion [ 11]. Also, the tensile strength is reduced, because the addition of ZrO2 particles impairs the binding force between PSF molecules.
Except for the manufacturing parameters mentioned above, the influence of pre-evaporation time and extraction temperature on the properties of ZF separator has been investigated in our previous paper [ 20]. The conclusion is that as pre-evaporation time prolongs, the maximum pore size, porosity and tensile strength are all decreased, and the corrosion resistance remained unaffected. However, the ionic resistance increases obviously when pre-evaporation time is more than 60 seconds. As extraction temperature increases, the maximum pore size decreases, and the corrosion resistance and the tensile strength remain unaffected, whereas the porosity firstly decreases and then increases with the inflexion at 45 °C.
ZF separators have numerous excellent properties, except for its vulnerability, for alkaline water electrolysis. Thus, it is necessary to strengthen its mechanical strength to cope with the demands of industrial application. So we prepared a modified separator with a support layer, which is composed of PPS felt with four different densities, because PPS felt has good mechanical stability and corrosion resistance to be used as support material.
The film casting solution is required to be exposed to the air for a certain time, so called pre-evaporation time, before the film is immersed into the non-solvent bath. In this process, moisture in the air will contact with the wet film and enable separating liquid phase to form a dense gel layer.
However, because the PPS felt has fluffy structure, it will expand during pre-evaporation and damage the dense gel layer to form large pores. As shown in Table 5, the pore size of ZF separator with the support layer is larger than that without the support layer obviously. Therefore, it is better to use thin felt to reduce deformation. As shown in Table 6, the maximum pore size decreases with the decrease of the felt thickness.
| Tab.5 The maximum pore size comparison between ZF and composite separators |
| Tab.6 The effect of the support layer thickness on the separator properties |
We also tested the tensile strength, and the results are presented in Table 6. It can been seen that the support layer greatly enhance the tensile strength of the composite separator, probably because the cast solution sufficiently infiltrate into the felt during immersion, and then the felt fiber bonds tightly due to the phase inversion, leading to great binding force. Table 7 also shows that with the increase of the felt density, the tensile strength is enhanced as a result of the increased amount of connective fibers.
The ionic resistance and resistivity of the separators with support layer is larger than those of ZF separator due to the hindering effect of fiber on the formation of cavernous structure.
| Tab.7 Comparison of the properties between ZF and composite separators |
In order to evaluate the practical applicability of those composite separators, a small-scale electrolysis experiment apparatus, as presented in Fig. 7, was applied to test the stability of the modified ZF separators and investigate the changes of cell voltage and gas purity as a function of current density.
1-cell electrolyzer, 2-hydrogen separator, 3-oxygen separator, 4, 5-circulating pump, 6,7-flow regulating valve, 8,9-flowmeter, 10-DC stabilized power supply, 11,12-heating rod, 13,14-condenser pipe, 15-cooling water circulation
A modified ZF separator with a support layer was applied in this small-scale electrolyzer. The electrolyte is KOH with concentration of 30 wt-% at 80 °C. Asbestos separator and ZF separator (300 μm) was submitted to the same tests for comparison. As shown in Fig. 8, the modified ZF separator has a smaller cell voltage than that of asbestos separator versus different current density, but the cell voltage of asbestos separator is higher than that of ZF separator due to the higher ionic resistance.
The gas purity was determined by an Orsat gas analyser using the traditional type of constant pressure volumetric method.
As shown in Table 8, H2 purity fluctuates with the changes of current density regardless of the separator used. However, both the composite separator and ZF separator have a higher H2 purity than the asbestos separator, which had been studied in our previous work and the H2 purity was determined to be from 99.3% to 99.5%. Therefore, the modified ZF separator has better gas-separation performance than the asbestos separator.
| Tab.8 The gas purity versus current density using different separators* |
Table 9 shows that the H2purity using modified ZF separator remains stable under electrolyzing from 5 to 15 days, indicating that the modified separator has a good chemical and thermal stability.
| Tab.9 Gas purity of different separators versus electrolyte time* |
We have demonstrated that the manufacturing parameters have great impacts on the properties of ZF separators. We also prepared a modified ZF separator by adding a support layer. This composite separator shows a dramatic increase from 1.85-13.66 MPa in the tensile strength. Compared with the conventional asbestos separator, this composite separator has better electrical conductivity and gas-separation property. With the excellent chemical, mechanical and thermal stability, the modified ZF separator may find application in industrial hydrogen production.
| 1 |
|
| 2 |
|
| 3 |
|
| 4 |
|
| 5 |
|
| 6 |
|
| 7 |
|
| 8 |
|
| 9 |
|
| 10 |
|
| 11 |
|
| 12 |
|
| 13 |
|
| 14 |
|
| 15 |
|
| 16 |
|
| 17 |
|
| 18 |
|
| 19 |
|
| 20 |
|